
We know from the periodic table that copper is in group 11, so this agrees with the result of this electron configuration calculator. Let us look at the electron configuration for helium, which is 1 s 2 1 4s^1 3 d 10 4 s 1. The electron configuration is responsible for many physical and chemical properties of an element. Ground state means that the atom has the lowest energy allowed. Since the 4s contains lower energy, the last electron of potassium which is the 19th electron must enter into the 4s orbital rather than the 3d orbital.Electron configuration notation provides us with information about the basic energy levels and sublevels that electrons occupy. Upon going from the noble-gas configuration of argon (atomic number 18) to potassium (atomic number 19), the added electron goes into the large 4s orbital. The last electron of the potassium should enter into the 3d orbital.ġ9K → 1s 2 2s 2 2p 6 3s 1 3p 6 3d 1 (but it does not happen)īut as we consider the energy of the 4s and 3d orbital by calculating the value of n+l, we can clearly notice that the n+l value of the 4s is lower than the 3d. The last electron of Potassium enters into 4s orbital – why? Therefore, though both 3d and 4p have the same n+l value, the energy of 4p is higher than 3d (3d<4p). The 4p orbital is in the fourth energy level, on the other hand, the 3d orbital is in the third energy level. In such a case the orbital in the higher energy level is considered to be contained higher energy. Therefore, according to the Aufbau principle, the 3d orbital should be fulfilled before the 4s orbital.įor 4p orbital- n=4 and l=p=1, therefore n+l = 5 Potassium is the alkali metal group with the electron configuration 2-8-8-1 or 1 s2 2 s2 2 p6 3 s2. Now, the 3d orbital is in the third energy level and the 4s orbital is in the fourth energy level. In Electronic Configuration electrons are arranged in various shells, Subshell and Orbital by following certain rules. That means at first, the electrons enter into the first energy level then the second energy level, the third and fourth energy level, and so on. Electron Configuration -The Electron Configuration of an Element Describes how Electrons are Distributed in their Atomic Orbitals. 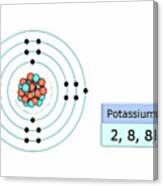
The Aufbau principle for the electronic configuration of the elements states that the electrons contained in an atom must fulfill the lower energy orbitals first. Therefore, the electronic configuration of this element is-ġ1Na→ 1s 2 2s 2 2p 6 3s 1 Aufbau principle For example, Na (Sodium) has eleven electrons. The Electronic configuration is the arrangement of the electrons in the orbitals. The electrons remain outside of the center (nucleus) of the atom in some energy levels and sub-energy levels. So what is electronic configuration? we all know that every substance is made of atoms as well as the atoms consists of the electron, protons, and neutrons. Electron Configuration of the elements Aluminum, Ne3s23p Mercury, Xe6s24f145d Americium, Rn7s25f Molybdenum, Kr5s14d Antimony, Kr5s24d105p. 
Electron configuration of Potassium is Ar 4s1. In the periodic table, the elements are listed in order of increasing atomic number Z. 
What is a quantum number what do quantum numbers determine? Electronic configuration The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. You can read the article below to get a clear view of it. First, you should have a clear knowledge of quantum numbers. To understand this fact properly, we should have a clear concept about some topic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
